For many emerging biotech and pharma companies, regulatory approval marks the beginning of a new layer of complexity rather than the end of one.
Labelling is one of the most consequential and most underestimated parts of launch readiness. Get it right early, and you protect your timeline. Leave it too late, or treat it as a final administrative step, and it can stall a drug development programme that has taken years to reach this point.
This blog breaks down the core EU and UK drug labelling requirements — from the Summary of Product Characteristics (SmPC) to patient-facing packaging — and explains why the marketing authorisation holder (MAH) carries ongoing responsibility for keeping labelling current, accurate and compliant across the product lifecycle.
What drug labelling actually covers
When most people think about drug labelling, they picture the outer box or the patient leaflet. In reality, it is a coordinated set of regulated documents and materials that together form the product’s approved information package.
In both the EU and UK, compliant labelling typically includes:
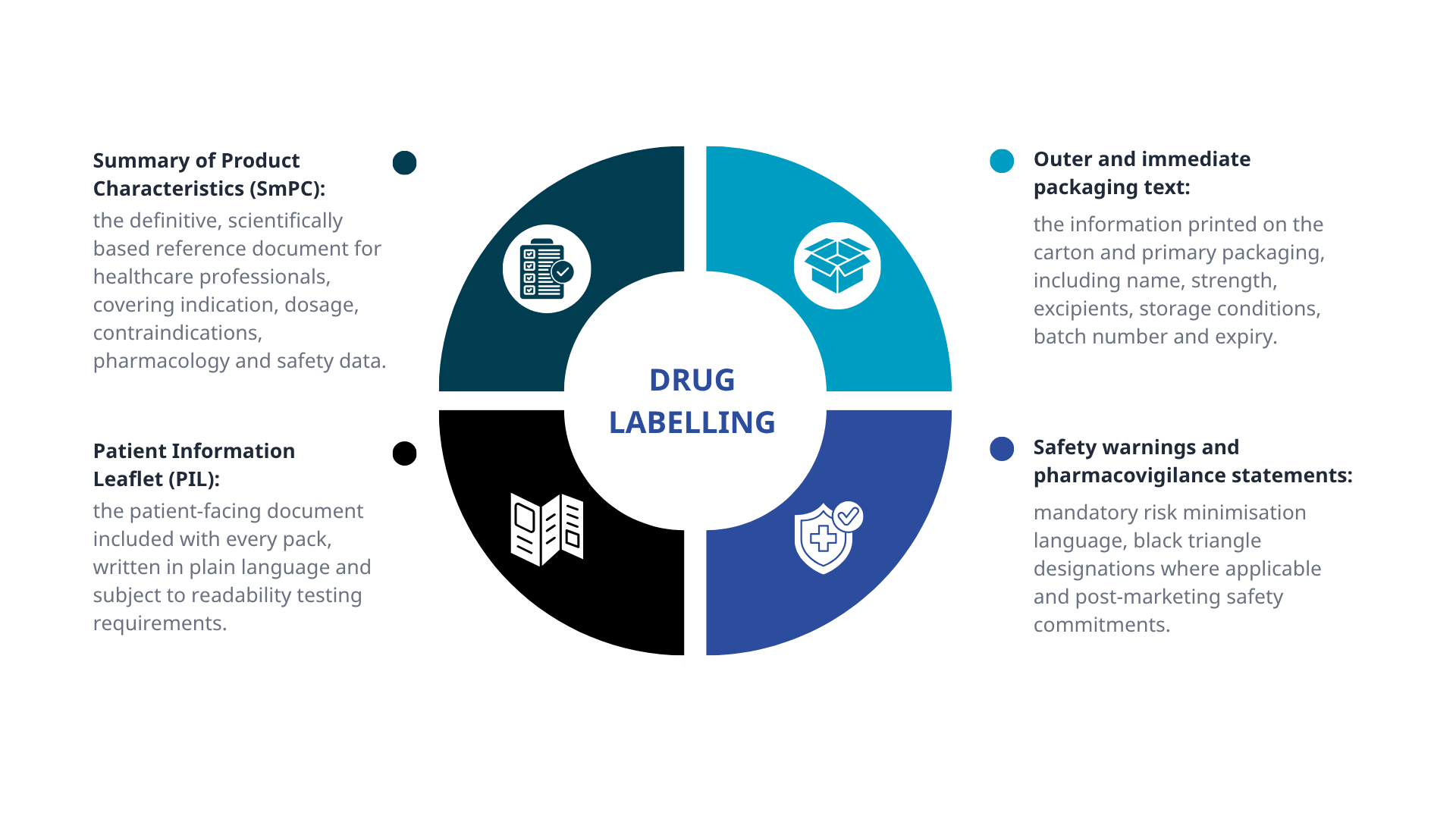
All of these elements must be consistent with the approved product information and updated whenever the regulatory authorities require it.
EU and UK frameworks: aligned in principle, distinct in practice
EU and UK drug labelling requirements share the same foundational principles — protecting patients, enabling informed clinical decision-making and ensuring transparency — but operate through separate regulatory systems with different processes, timelines and templates.
EU labelling is governed by the European Medicines Agency (EMA) and must conform to the Quality Review of Documents templates, which specify the structure, language and content requirements for each document type. Submissions go through the centralised procedure for medicines approved across EU member states, or through national, decentralised and mutual recognition procedures for those seeking approval in individual countries.
In the UK, the Medicines and Healthcare products Regulatory Agency (MHRA) oversees labelling requirements. The MHRA has its own product information templates and submission processes. While these mirror many EU conventions, there are important divergences — particularly around terminology, reference safety information and the handling of bilingual labelling for products intended for both Great Britain and Northern Ireland.
For sponsors seeking access in both markets, this means maintaining two sets of parallel approved labelling: each aligned to its own regulatory authority, each subject to its own variation procedures when updates are needed. Working with a specialist pharma services company that understands both systems is an effective way to manage this complexity without building duplicate internal capability.
Drug labelling requirements in both regions are also not static. The EU’s ongoing pharmaceutical reform — with significant provisions rolling out from 2026 — is expected to strengthen transparency obligations, expand digital labelling options and increase scrutiny of product information for complex or novel therapies. You need to account for this evolving landscape from the outset, when planning a dual EU/UK launch.
For global licences, consistency across other countries, such as the US, is also paramount. All regional labels must be traceable back to the Company Core Data Sheet as the global source of truth.
Why drug labelling needs to be part of clinical development planning
One of the most common mistakes first-time sponsors make is treating labelling as a submission task rather than a clinical development consideration. In practice, the language in your SmPC is directly shaped by the evidence you generate and the claims your programme supports. If your labelling strategy is not integrated into protocol design and evidence planning early, you risk reaching the end of development with data that does not clearly support the product information you need.
This is particularly acute for advanced therapy medicinal products, such as cell and gene therapies. These products often involve small patient populations, conditional or accelerated approval pathways, and complex safety and handling requirements that are difficult to describe in standard templates. The more novel the therapy, the more important it is to start labelling planning before the first data package is compiled.
Accelerated pathways — such as a conditional marketing authorisation — add further complexity. When approval is granted on the basis of less complete data, the labelling must reflect this accurately, including any conditions attached to the authorisation and ongoing commitments to generate additional evidence. This means drug labelling may need to evolve more rapidly than for a product approved through a standard pathway.
Digital tools and electronic submission systems are also raising the bar for accuracy and consistency. Errors that might once have been caught late in a manual review process are now more likely to generate questions or rejection at the point of regulatory submission — putting greater emphasis on document governance and version control throughout clinical development.
Why cross-functional alignment is critical to avoid drug labelling delays
Even when the regulatory knowledge is in place, labelling delays frequently originate in a different problem: disconnected teams.
The information that goes into product labelling draws on clinical data, pharmacovigilance outputs, medical affairs input, regulatory strategy, quality review and commercial artwork management. When these functions operate independently, with different timelines and no shared governance for labelling decisions, inconsistencies accumulate. A safety update agreed by pharmacovigilance may not be reflected promptly in the SmPC. An artwork change initiated by commercial teams may not have gone through the correct regulatory approval process.
This is one of the most common patterns seen in emerging companies managing their first EU or UK approval. The solution is not simply more resource — it is a coordinated approach with clear ownership, defined processes and a single point of accountability for labelling across the lifecycle. An experienced pharma services company can provide exactly that structure, integrating labelling governance into your broader programme management from the start.
Labelling doesn’t stop at approval: the marketing authorisation holder’s ongoing responsibilities
Understanding drug labelling requirements is not only about getting to approval — it is about maintaining compliance over the product’s entire commercial life. This is where the role of the marketing authorisation holder becomes critical.
The MAH is the legal entity responsible for the product in each market. In the context of drug labelling, this means the MAH must:
- Monitor safety literature and pharmacovigilance data and initiate labelling updates when new safety signals emerge.
- Submit variations to the EMA and/or MHRA when the product information needs to change — whether due to safety findings, new indications, updated dosing guidance or manufacturing changes.
- Ensure that updated labelling is implemented across all packaging artwork, translations and pharma access markets within the timelines set by the regulators.
- Maintain accurate, inspection-ready documentation of all labelling versions, change histories and artwork approvals.
- Coordinate the end-to-end artwork process: from briefing the design function through to regulatory review, print readiness and distribution.
This is a significant operational undertaking. A single variation can involve multiple document updates, translations across EU member states, cross-functional review cycles and printer file management — all running to regulatory deadlines. For a product with a broad geographic footprint or a complex safety profile, the volume of labelling activity over the lifecycle can be substantial.
Labelling non-compliance — whether through missed safety updates, unapproved artwork changes or documentation gaps — can result in regulatory action, product recalls, inspection findings or damage to the market authorisation itself, so getting it right is crucial.
How an outsourced marketing authorisation holder reduces the operational burden
For many emerging biotech and pharma companies, the answer to this operational complexity lies in partnering with an experienced outsourced MAH. Rather than building the infrastructure internally — which requires significant investment in regulatory expertise, quality systems, pharmacovigilance capability and labelling management processes — outsourcing allows you to access that infrastructure from day one.
A specialist pharma services company acting as your marketing authorisation holder in the EU and/or UK takes on the legal and operational responsibilities associated with the authorisation, including labelling lifecycle management. This means handling variation submissions, coordinating artwork updates, managing translations, maintaining compliant documentation and providing the cross-functional oversight that keeps drug labelling current across pharma access markets.
The best outsourced MAH partners work as an extension of your team, giving you visibility and control over your product while managing the day-to-day regulatory and operational workload that would otherwise fall on internal resource that many emerging companies simply do not have.
For first-time sponsors in particular, this model offers a meaningful advantage: you retain ownership of your asset and your commercial strategy while a specialist drug commercialization solution manages the compliance infrastructure. That distinction matters — especially if you are evaluating whether to establish your own legal entity in the EU or UK, or whether a longer-term outsourced arrangement better suits your stage of development and resource base.
Outsourcing also gives you flexibility. A drug commercialization solution can be a bridging arrangement while you build internal capability, or a long-term operating model if the economics and risk profile support it. Either way, it allows you to compress the path to market authorisation and launch without compromising on the regulatory rigour that protects your product once it is in market.
Getting drug labelling right from the start
Drug labelling requirements in the EU and UK are detailed, dynamic and consequential. They connect directly to clinical evidence, regulatory strategy, pharmacovigilance and commercial operations — which is why a siloed or reactive approach rarely works.
The sponsors who manage labelling most effectively tend to share a few characteristics: they start planning early, they integrate labelling considerations into their clinical and regulatory strategy, and they have clear governance and accountability across the functions that contribute to it.
For those who do not yet have that infrastructure in place — or who are approaching their first EU or UK launch — the right partner can make a significant difference to both the speed and the confidence of market entry.
TMC Commercial provides flexible, comprehensive EU and UK drug commercialization solutions — from preparing for regulatory submission through to post-authorisation lifecycle management — giving you the opportunity to build your own commercial presence without needing to out-license to a partner. This includes the expertise to act as your marketing authorisation holder, managing labelling obligations, variation submissions and artwork governance on your behalf.
We help you make informed, long-term decisions that reduce the time, cost and operational complexity associated with market entry — so you retain control of your asset without the burden of building the infrastructure from scratch. To find out how TMC Commercial can support your EU or UK launch, contact our team at connect@tmcpharma.com.
