Remote engagement is no longer a ‘nice to have’ in clinical research — particularly when you’re working with small, geographically dispersed patient populations, intensive assessment schedules or highly specialised sites.
But introducing home visits, telehealth, local healthcare providers (HCPs) and digital endpoints into early-phase clinical trials changes the risk profile overnight.
Early studies define the safety profile, establish pharmacokinetics/pharmacodynamics and inform go/no-go decisions across clinical development. In rare disease and complex oncology settings, the stakes are even higher: cohorts are small, participants are often medically vulnerable, and each datapoint can meaningfully influence how regulators and investors perceive benefit–risk.
This is where clinical governance must become deliberate, structured and technology-aware — not simply a traditional monitoring plan ported into a hybrid model.
Below, we outline practical controls and monitoring approaches to maintain governance when implementing decentralised elements — with specific considerations for clinical trials for rare diseases, complex oncology and advanced therapeutic modalities.
Why decentralised clinical trials are reshaping clinical development in specialist indications
Decentralised elements can make participation feasible for patients who might otherwise be excluded due to travel limitations, disability, caregiver burden or distance from specialist centres.
A core reality of clinical trials for specialist indications is that patients often face long diagnostic journeys and may be under the care of a small number of specialist centres — sometimes hundreds of miles away. Hybrid and decentralised approaches can help bring selected trial activities closer to the participant while preserving specialist oversight.
The same benefits apply across complex settings:
- Rare disease: small, dispersed populations and reliance on specialist centres make hybrid participation models essential for feasibility.
- Complex oncology: frequent assessments and participant frailty can increase dropout risk; reducing travel and caregiver burden supports retention and continuity of safety monitoring.
- Advanced therapeutics: highly specialised administration may remain site-based, but follow-up assessments and patient monitoring can often be decentralised — reducing burden without compromising the core intervention pathway.
Decentralised clinical trials (where trial activity occurs remotely or in non-traditional settings) and hybrid models can, therefore, support:
- Access and recruitment: broader geographic reach for small patient populations, including rare oncology diseases.
- Retention and protocol feasibility: fewer exhausting site visits after long travel, which is particularly important when the disease burden is high.
- Timelier safety insights: remote data can enable faster review and escalation when appropriately governed.
Regulators are also now more explicit about expectations. The FDA’s final guidance on conducting trials with decentralised elements describes recommendations for sponsors and investigators on design, conduct and oversight — reinforcing that decentralisation is acceptable when controls are fit-for-purpose.
However, it’s important to note that decentralisation doesn’t reduce sponsor or investigator responsibilities. It redistributes them across more people, places and technologies — and that is exactly where governance makes or breaks the model.
Practical governance controls for decentralised clinical trial elements
When implemented with strong governance, decentralised elements can support the operational feasibility of ambitious early-phase clinical trials while maintaining the confidence needed for later-stage investment and regulatory dialogue.
In hybrid and decentralised clinical trials, clinical governance should do five things consistently:
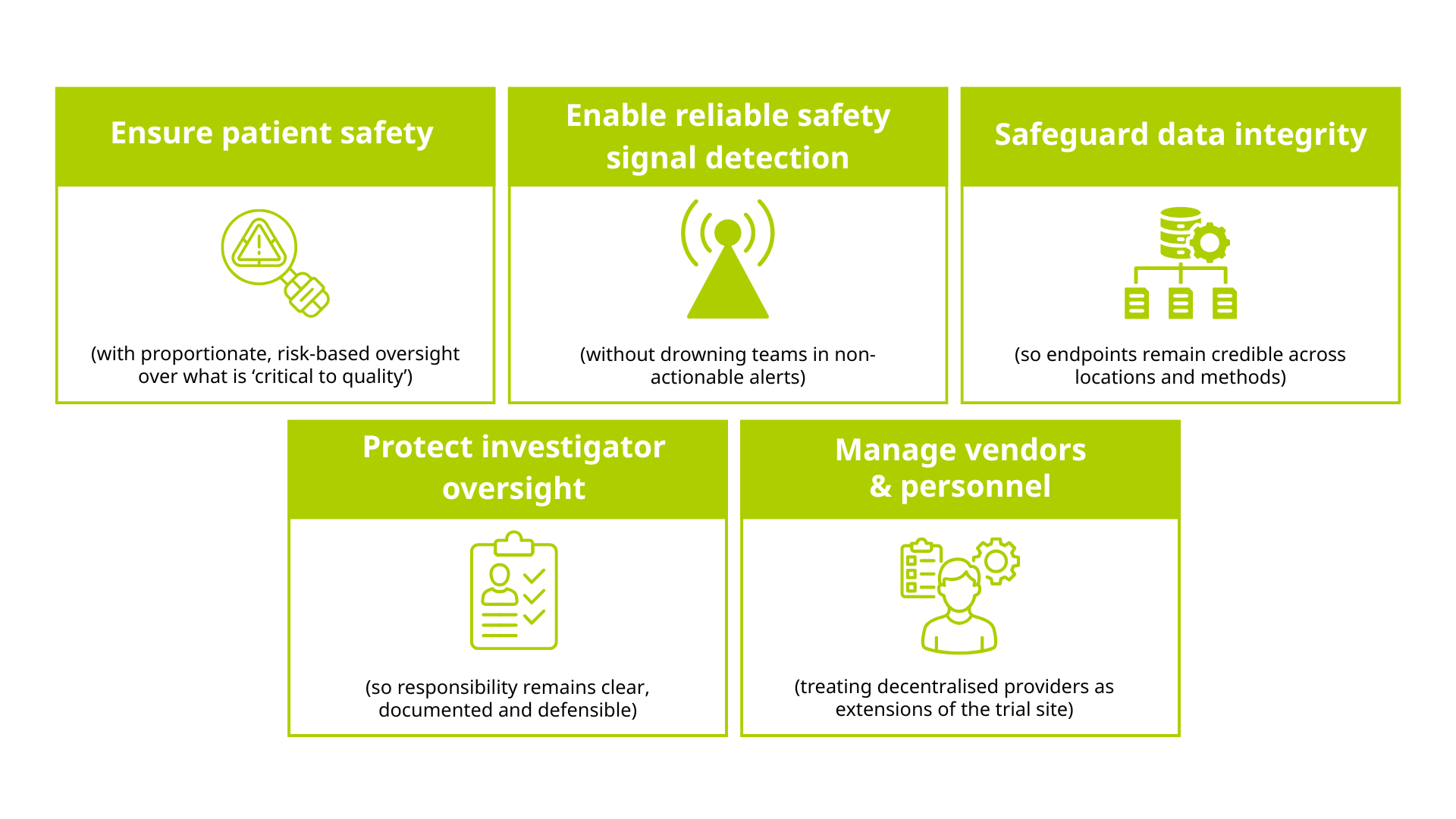
For rare disease, complex oncology and advanced therapeutics, this risk focus is essential. Small populations and complex causality (e.g. disease progression vs drug-related events) can make safety interpretation harder, while first-in-human risk can evolve rapidly — demanding real-time safety review and clear stop rules.
1. Define investigator oversight in a decentralised operating model
The first control is clarity: who does what, where, under whose supervision, and how that supervision is evidenced.
Practical steps include:
- Creating a RACI (Responsible, Accountable, Consulted, and Informed) document, or Responsibility Assignment Matrix, that links each decentralised procedure (eConsent, home nursing, remote vitals, local lab draws, telehealth) to:
- responsible role (investigator, delegated site staff, vendor staff, local HCP),
- oversight mechanism (real-time review, scheduled review, sign-off, committee review),
- documentation location (Trial Master File, vendor portal, EDC).
- Updating delegation logs to reflect remote personnel and specify limits of responsibility (what they can do versus what must return to the investigator).
- Ensuring remote access rights support oversight (e.g. investigator access to dashboards, source data views where appropriate and audit trails).
This is a foundational clinical governance requirement: when activities move off-site, accountability must not become ambiguous.
2. Build safety signal detection around escalation pathways
Safety detection improves only when the system converts data into timely clinical decisions.
A robust approach includes:
- Pre-defined thresholds and triggers for labs, vitals, wearable-derived measures and patient-reported outcomes — aligned to protocol stop rules.
- Tiered escalation so that:
- low-risk variances route to the study team for follow-up,
- clinically significant triggers route to the investigator/medical monitor immediately,
- persistent or clustered signals prompt committee review (e.g. safety review meeting cadence).
- Medical monitoring integration: ensure medical oversight is not an afterthought in hybrid delivery models. Remote medical monitoring should be part of integrated medical services supporting study safety and benefit-risk documentation.
In rare disease and oncology, this is particularly important because causality can be nuanced, and each serious adverse event can disproportionately influence perceived risk. Without disciplined clinical governance, alert fatigue, inconsistent follow-up and delayed causality assessment become likely failure points.
3. Standardise data flows to protect integrity across multiple sources
Hybrid trials often create data sprawl: multiple vendors, devices and portals generating parallel datasets.
Controls that reduce integrity risk include:
- Single source of truth for endpoint data, defining what ultimately populates electronic data capture (EDC) and supports analysis.
- Data flow mapping (from device/vendor to ingestion to cleaning to review to database lock), including where audit trails sit.
- Central review routines: scheduled data review sessions that focus on critical data, missingness and cross-source consistency.
- Computer system validation and vendor qualification (especially for digital health technologies and eConsent), with documented change control and version tracking.
This is where clinical governance intersects directly with inspection readiness — because regulators will expect you to demonstrate not only what you did, but how you ensured reliability.
4. Use risk-based monitoring that reflects decentralised reality
Traditional monitoring does not scale to deliver decentralised clinical trials. Instead, align monitoring intensity to critical-to-quality factors (participant safety and primary endpoints), as the latest GCP expectations encourage.
A practical risk-based model might prioritise:
- eligibility confirmation Dosing, and key safety assessments (early-phase).
- Endpoints that are susceptible to variability when performed at home versus site.
- Sites/patients with higher protocol deviation risk.
- Technology-enabled endpoints requiring verification.
For clinical trials for rare diseases, complex oncology and advanced therapeutics, this approach is particularly valuable because you often need flexibility to keep participation feasible — but cannot afford ambiguity in critical data.
5. Treat decentralised personnel and vendors as part of your quality system
Home nurses, mobile phlebotomy, local imaging centres, device vendors and call centres are effectively extensions of the trial site. The governance question is how to oversee and manage these vendors.
Key controls include:
- Documented qualification and training (role-specific, protocol-specific and escalation-specific).
- Competency checks for procedures that influence endpoints.
- Communication pathways for urgent safety issues (24/7 coverage where appropriate).
- Reconciliation plans (e.g. local lab reports versus EDC entries; home visit notes versus source).
These controls become even more critical in advanced therapeutic modalities, where site capability, handling procedures and adverse event readiness can be highly specialised. In clinical trials for rare diseases, selecting sites with the right infrastructure (e.g. cellular therapy suites) and investing in training for product handling and adverse-event management is essential.
Supporting governance-led decentralisation in early-phase clinical trials
Any study-specific governance model should be aligned to your protocol, risk assessment and applicable regulations.
If you’re introducing decentralised clinical trial elements into your next programme and want a governance-led approach that protects safety, data integrity and oversight, TMC can help you design the operating model, controls and monitoring pathways to make it work.
TMC Clinical specialises in early-phase clinical trials and full-service clinical development solutions for rare disease, complex oncology and advanced therapeutic modalities — combining the responsiveness of a specialist pharma services company with the expertise of a clinical research organisation.
Across early-phase clinical development, TMC’s support spans site feasibility, key opinion leader identification, clinical trial application submissions, ongoing site management, medical monitoring and reporting to relevant regulatory gateways.
Speak to us today at connect@tmcpharma.com to discuss your programme and how we can support your next early-phase trial in rare disease, complex oncology or advanced therapeutic modalities.
